1. SUMMARY
In February 1992, the National Institute for Occupational Safety and Health (NIOSH) received a request for a health hazard evaluation (HHE) from a management representative of the Federal Bureau of Investigation (FBI) in Washington, D.C. The request was submitted because of concern about possible health hazards due to the exposure of fingerprint specialists to many of the chemicals that they use in processing latent fingerprints. NIOSH evaluated potential exposures to ethyl-2-cyanoacrylate, petroleum ether, iodine, acetone, titanium dioxide, and carbon black in the fingerprint laboratories and at a simulated crime scene. The laboratories were equipped with exhaust hoods, but there was no exhaust ventilation available at the simulated crime scene. NIOSH investigators also evaluated an argon laser that was used by fingerprint specialists to examine objects for visible fingerprints.
On July 30-31, 1992, NIOSH investigators conducted a walk-through evaluation of the Latent Fingerprint Section (LFPS) of the FBI Identification Division and on October 27-28, 1992, air samples were collected in the LFPS laboratories while fingerprint specialists developed latent fingerprints in the laboratory exhaust hoods. These air samples were analyzed for airborne iodine, petroleum ether, ethyl-2-cyanoacrylate, and acetone. None of the airborne concentrations of any of these compounds were above environmental or occupational exposure criteria established by NIOSH, the Occupational Safety and Health Administration (OSHA), or the American Conference of Governmental Industrial Hygienists. The airborne concentration of ethyl-2-cyanoacrylate in the lab was 0.29 parts per million (ppm). The highest airborne concentration of petroleum ether to which fingerprint specialists were exposed in the lab was 52.4 milligrams per cubic meter (Mg/M3), less than the NIOSH Recommended Exposure Limit (REL) of 350 Mg/M3 . Air sampling for iodine yielded only one sample that contained a detectable amount of iodine. That sample was collected in the exhaust hood. Similarly, the only sample on which acetone was detected was collected in the exhaust hood. Air sample time in the laboratory ranged from 20 minutes to 141 minutes; all concentrations reported are averaged over the duration of the sample period.
On February 24, 1993, NIOSH investigators collected air samples at a simulated crime scene that was prepared by the FBI. This air sampling
survey included analysis for airborne iodine, petroleum
ether, ethyl-2cyanoacrylate, and fingerprint powders. Workers used
synthetic, chemical-protective gloves while using these materials to prevent
dermal contact; proper respiratory protection was not used. Airborne
personal breathing zone (PBZ) ethyl-2-cyanoacrylate concentrations at the
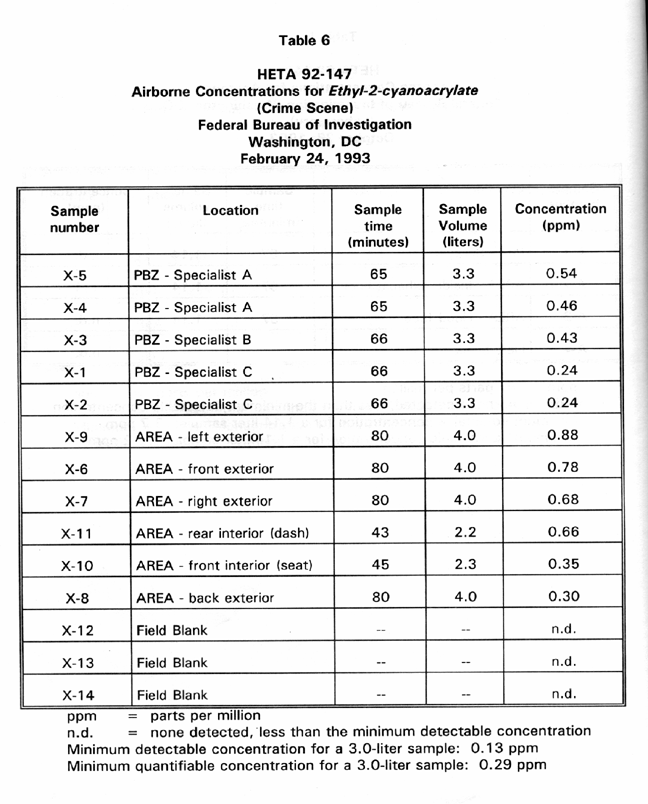
simulated crime scene ranged from 0.24 to 0.54 ppm, with a mean of 0.38
ppm. Concentrations in area samples ranged from 0.30 to 0.88 ppm,
with a mean of 0.61 ppm. Airborne PBZ petroleum ether concentrations
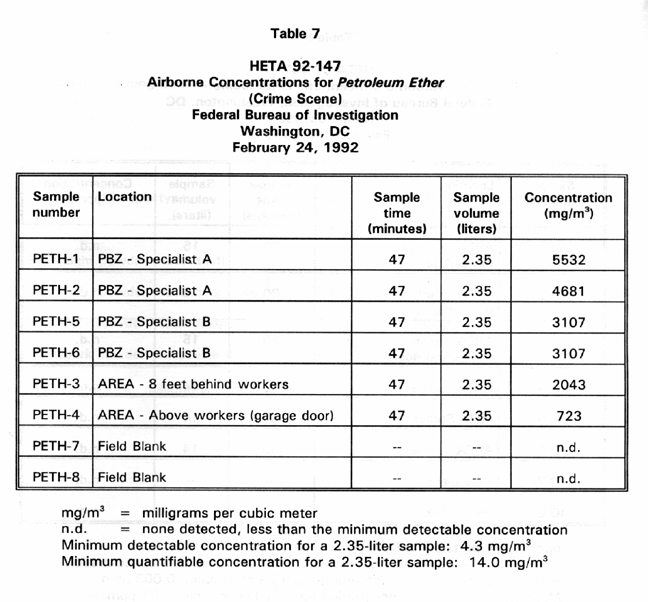
ranged from 3107 to 5532 mg/m' with a mean of 4107 Mg/M3 . Although the
sample duration for the highest PBZ petroleum ether concentration was only
47 minutes, the 10-hour time-weighted average for this sample exceeded
the NIOSH REL of 350 Mg/M3 . Analysis of two area samples for petroleum
ether revealed concentrations of 723 and 2043 mg/m'. Sample time
at the simulated crime scene ranged from 14 minutes to 80 minutes; concentrations
reported are averaged over the duration of the sample period.
Based on the results of this evaluation, it was determined
that the fingerprint specialists' chemical exposures while processing latent
fingerprints at crime scenes could be a potential health hazard.
Recommendations to address this hazard, including the use of respiratory
protection while using ninhydrin, iodine, and dark and light fingerprint
powders containing carbon black and titanium dioxide, respectively, are
presented in Section VIII of this report.
KEYWORDS: SIC 9221 (Police Protection), ethyl-2-cyanoacrylate, superglue, iodine, petroleum ether, acetone, fingerprint powder, titanium dioxide, carbon black, argon laser, latent fingerprints
Page 3 - Health Hazard Evaluation Report No. 92-147
II. INTRODUCTION
In February 1992, the National Institute for Occupational Safety and Health (NIOSH) received a request for a health hazard evaluation (HHE) from a management representative of the Federal Bureau of Investigation (FBI) in Washington, DC. The request was made because of employee complaints of respiratory problems and concern regarding exposure to various chemical compounds which are used by FBI fingerprint specialists to develop latent fingerprints. These fingerprint specialists worked in the Latent Fingerprint Section (LFPS) of the FBI Identification Division.
On July 30-31, 1992, NIOSH investigators conducted a walk-through evaluation of the LFPS. This site visit included visual observation of the many methods that the fingerprint specialists used to process latent fingerprints. On October 27-28, 1992, NIOSH investigators collected air samples in the LFPS laboratories while fingerprint specialists developed latent fingerprints in the laboratory exhaust hoods. These air samples were analyzed for iodine, petroleum ether, ethyl-2-cyanoacrylate, and acetone. On February 24, 1993, NIOSH investigators collected air samples at a simulated crime scene that was prepared by the FBI to simulate their working conditions at crime scenes. At the simulated crime scene, two fingerprint specialists employed some of the same fingerprint processing methods that were used in the laboratory. Air samples collected during this survey were analyzed for iodine, petroleum ether, ethyl-2-cyanoacrylate, and fingerprint powders.
An interim letter dated September 22, 1993, presented the results of the iodine portion of this investigation. Information contained in that letter has been incorporated into this report.
Ill. BACKGROUND
The FBI headquarters, located at 10th Street and Pennsylvania Avenue in Washington, DC, is an 11-story building containing offices and laboratories. The LFPS, which occupies a portion of one floor, consists of office space along the perimeter of the building with several laboratories toward the interior of the building. The LFPS is primarily responsible for the examination of irreplaceable crime scene evidence in order to locate, preserve, and identify latent prints. Location of latent prints is accomplished by visual examination or through the application of fingerprint powders, chemical developers and enhancers, lasers, and other light sources. Preservation of latent prints is accomplished through photography and/or lifting with transparent or rubber lifting tape designed for this purpose.
Page 4 - Health Hazard Evaluation Report No. 92-147
Fingerprint specialists use the laboratories to develop latent fingerprints from pieces of physical evidence that are sent to the LFPS from FBI field offices or from police departments nationwide. There are several chemical processes that can be used to develop latent prints, depending on the type of evidence at hand. The specialists mix the chemicals in the laboratory and use a spray gun to apply the chemicals to the evidence. This is done in the exhaust hoods. After the chemically-treated evidence is allowed to dry in the hoods, it is taken to the specialist's desk for further visual examination. These pieces of evidence are stored in large metal cabinets located throughout the section.
One of the methods used by FBI fingerprint specialists involves the use of superglue, which is composed primarily of ethyl-2-cyanoacrylate. In the laboratory, after placing the evidence in a cabinet, several drops of superglue are added to a small aluminum dish, which is then placed inside the cabinet. The superglue is then heated using a hot plate, and the vapors that are created will eventually condense on the fingerprints, allowing them to be easily seen and photographed. The specialists allowed the evidence to be exposed to superglue vapors for about five minutes before the vapors were exhausted from the cabinet into the laboratory exhaust hoods. At the simulated crime scene, large sheets of plastic were used to cover the automobile that was processed. The plastic was used to contain the superglue vapors that were created under the plastic. After several minutes, the plastic was removed and the vapors were allowed to dissipate. No local exhaust ventilation was available at the simulated crime scene.
The fingerprint specialists also go to crime scenes to process latent fingerprints. On these occasions, they often work in excess of 10 hours a day to fully process the crime scene. At the crime scene, it is usually not feasible to limit exposures to airborne contaminants via local exhaust ventilation.
At the time of this investigation, the smoking policy at the FBI headquarters allowed employees to smoke in designated smoking rooms and in some restrooms. The smoking area nearest the LFPS did not have a dedicated exhaust system at the time of this investigation.
IV. EVALUATION DESIGN AND METHODS
During the laboratory evaluation, six area air samples were collected to determine airborne concentrations of ethVI-2-cVanoacrViate, the main component of superglue. Several fingerprint specialists used a large metal cabinet to develop fingerprints using superglue. Personal breath zone (PBZ)
Page 5 - Health Hazard Evaluation Report No. 92-147
samples were not collected in the laboratory because each specialist used the cabinet for only a few minutes. During the simulated crime scene evaluation, both area and PBZ air samples were collected. During this evaluation, three fingerprint specialists used a large plastic cover to enclose on automobile, which they then exposed to superglue vapor for about 1 0 minutes. They removed the plastic cover and then left the room for several minutes to allow the vapors to clear. They also exposed the car interior to superglue vapors. PBZ samples were collected from these three specialists, and area air samples were collected in and around the automobile during the entire process.
Airborne ethyl-2-cyanoacrylate concentrations were measured using solid sorbent tubes (treated XAD-7) connected via Tygon' tubing to battery-powered vacuum pumps calibrated at flow rates ranging from 0.02 to 0.1 liters per minute (Lpm). These sorbent tubes were analyzed using high performance liquid chromatography (HPLC) using Occupational Safety and Health Administration (OSHA) method 55.' The analytical limit of detection (LOD) for the samples collected in the laboratory was 1.0 microgram (ug) per sample, which equates to a minimum detectable concentration (MDC) of 0.03 parts per million (ppm) for a 7-liter sample. The analytical limit of quantitation (LOQ) for the samples collected in the laboratory was 3.3 /ug per sample, which equates to a minimum quantifiable concentration (MQC) of 0.09 ppm for a 7-liter sample. The LOD for the samples collected at the simulated crime scene was 2.0 /ug per sample, which equates to a MDC of 0. 1 3 ppm for a 3-liter sample. The LOQ for the samples collected at the crime scene was 4.5 /ug per sample, which equates to a MQC of 0.29 ppm for a 3-liter sample.
A chemical called ninhydrin is widely used to develop latent fingerprints. There are no available air sampling methods to measure airborne ninhydrin concentrations, and no environmental or occupational exposure criteria to evaluate the relative hazard of airborne ninhydrin concentrations. The carrier for ninhydrin used by the LFPS is petroleum ether, which is easily measured and for which occupational exposure guidelines exist. Because of this, airborne concentrations of petroleum ether were measured in the lab and at the crime scene when the specialists used the ninhydrin method. These measurements were made to determine how much greater the exposure was at the crime scene compared to the laboratory when the specialists used the ninhydrin solution to develop latent fingerprints.
Airborne petroleum ether concentrations were measured using solid sorbent tubes (1 50 milligrams [mg] of charcoal) connected via Tygon' tubing to battery-powered vacuum pumps calibrated at either 0.02 or 0.05 lpm.
Page 6 - Health Hazard Evaluation Report No. 92-147
These charcoal tubes were analyzed using gas chromatography (GC) and a flame ionization detector (FID) according to NIOSH method 1550.' The LOD
for these samples was 0.01 mg per sample. The MDC for the laboratory
samples was 16.7 Mg/M3 for a 0.6-liter sample, and 4.3 mg/m' for a
2.35-liter sample at the crime scene. The LOQ for these samples was
0.033 mg per sample, which equates to MQCs of 55. 0 Mg/M3
for a 0.6-liter sample (laboratory), and 14.0 mg/M3 for a 2.35-liter sample
(crime scene).
An iodine solution was also used by the fingerprint specialists
in the laboratory and at the crime scene. PBZ and area samples were
collected at both sites. Airborne iodine samples were collected on
solid sorbent tubes (1 50 mg of alkali-treated activated charcoal) connected
via Tygon' tubing to battery-powered vacuum pumps calibrated to flow rates
of 0.5 and 1.0 lpm. The sorbent tubes were analyzed using ion chromatography
according to NIOSH method 6005. The LOD for the laboratory samples
was 1.1 jig per sample, which equates to a MDC of 0.0005 ppm for a 20-liter
sample. The LOQ for the laboratory samples was 3.7 /ug per sample,
which equates to a MQC of 0.02 ppm for a 20-liter sample. The crime
scene LOD was 0.4,pg per sample, which corresponds to a MDC of 0.003 ppm
for a 15-liter sample. The LOQ for the crime scene samples was 1.3
/ug per sample, which equates to a MQC of 0.01 ppm for a 15-liter sample.
Another solution that is used to develop prints is called DFO (1,8-diazofluoren-9-one). Acetone is a component of the DFO mixture. PBZ and area air samples were collected in the laboratory to measure the airborne acetone concentration while DFO was used to spray evidence. Airborne acetone concentrations in the laboratory were collected on charcoal tubes connected via Tygon' tubing to battery-powered vacuum pumps calibrated at a flow rate of O..02 lpm. The charcoal tubes were analyzed using GC/FID according to NIOSH method 1300. The LOD was 0.01 mg per sample, which equates to a MDC of 3.7 ppm for a 1.14-liter sample. The LOQ was 0.033 mg per sample, which equates to a MQC of 12.2 ppm
for a 1.14-liter sample.
At the simulated crime scene, PBZ air samples were collected to determine the airborne concentrations of "light" and "dark" fingerprint powders used by the fingerprint specialist. These air samples were collected on tared 37-mm polyvinyl chloride filters connected via Tygon'" tubing to battery-powered vacuum pumps calibrated at flow rates of 1.7 and 2.0 lpm. Samples were analyzed gravimetrically according to NIOSH method 0500 According to the Material Safety Data Sheets (MSDSs), the light powder contained titanium dioxide and the dark powder contained carbon black.
Page 7 - Health Hazard Evaluation Report No. 92-147
In addition to the air samples that were collected, a
visual assessment of the effectiveness of the laboratory exhaust hoods
was made.
A smoke-generator was used to demonstrate air movement
in and around the exhaust hoods. The smoke generator was also used
to determine the relative pressure differential between the laboratories
and the rest of the LFPS offices. The locations of the area air samples
that were collected in the laboratory during the superglue process were
based on the results of the visual assessment of air movement in the laboratory.
NIOSH investigators also evaluated the use of the two argon lasers that were used to help detect latent fingerprints on the surface of items prior to chemical processing. The NIOSH evaluation of the lasers consisted of direct observation of the lasers and the safety equipment available, and an interview of the laser operator.
V. EVALUATION CRITERIA
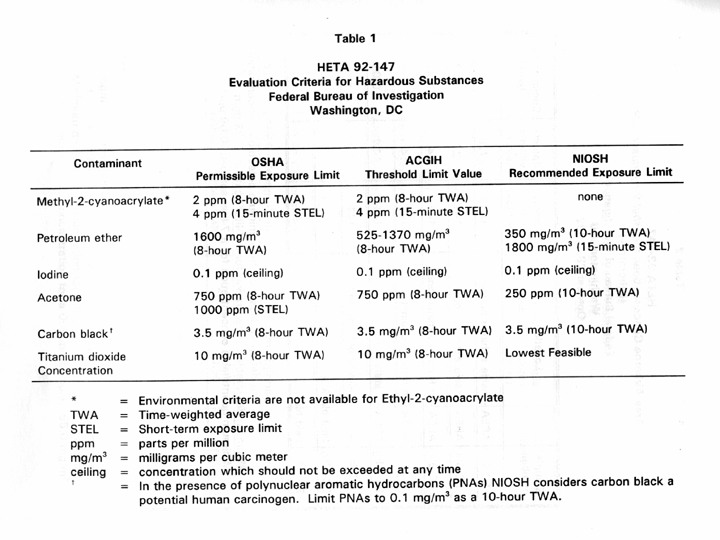
As a guide to the evaluation of the hazards posed by workplace exposures, NIOSH field staff employ environmental evaluation criteria for the assessment of a number of chemical and physical agents. These criteria are intended to suggest limits of exposure to which most workers may be exposed up to 1 0 hours per day, 40 hours per week for a working lifetime with-out experiencing adverse health effects. It is, however, important to note that not all workers will be protected from adverse health effects even though their exposures are maintained below these limits. A small percentage may experience adverse health effects because of individual susceptibility, a pre-existing medical condition, and/or a hypersensitivity (allergy). In addition, some hazardous substances may act in combination with other workplace exposures, the general environment, or with medications or personal habits of the worker to produce health effects even if the occupational exposures are controlled at the limit set by the criterion. These combined effects are often not considered in the evaluation criteria. Also, some substances are absorbed by direct contact with the skin and mucous membranes, and thus potentially increase the overall exposure. Finally, evaluation criteria may change over the years as new information on the toxic effects of an agent become available.
The primary sources of environmental evaluation criteria for the workplace are the following: (1) NIOSH Recommended Exposure Limits (RELs),' (2) the American Conference of Governmental Industrial Hygienists (ACGIH) Threshold Limit Values (TLVs), and (3) the U.S. Department of Labor, OSHA Permissible Exposure Limits (PELs).' The OSHA PELs may be required to take into account the feasibility of controlling exposures in various industries
Page 8 - Health Hazard Evaluation Report No. 92-147
where the agents are used; the NIOSH RELS, by contrast, are based primarily on concerns relating to the prevention of occupational disease. In evaluating the exposure concentrations and the recommendations for reducing these concentrations found in this report, it should be noted that the lowest exposure criteria was used; however, employers are legally required to meet those limits specified by the OSHA standard.
A time-weighted average (TWA) exposure refers to the average airborne concentration of a substance during a normal 8- to 10-hour workday. Some substances have recommended short-term (1 5-minute average) exposure limits (STELS) or ceiling values (not to be exceeded at any time) which are intended to protect workers from recognized toxic effects of high short-term exposures. The evaluation criteria for the chemicals investigated during this evaluation are presented in Table 1.
Ninhydrin
Ninhydrin is a triketone (1,2,3-inadanetrione) which is
primarily used in the fields of analytical chemistry, biochemistry, and
forensic science.
Ninhydrin's ability to react with amino groups enables
its use as a latent fingerprint reagent. The ability to react with
amino groups, including amino acids which are the "building blocks" of
proteins, have prompted concerns about exposure and toxicity.
A review of available literature revealed few sources of information on ninhydrin toxicity. An experiment with mice was reported in 1957; it assessed lethal and toxic dose levels for direct intraperitoneal and intravenous injection of ninhydrin. The mechanism of toxicity was not established.9 Ninhydrin's structural similarity to alloxan, which is diabetogenic, prompted speculation that ninhydrin, too, can cause diabetes. 9 However, this hypothesis has not yet been tested.
Two reports in the literature describe ninhydrin-induced allergic contact eczema. One report recounts the reactions of three individuals to the ninhydrin component of ink found in a surgical marking pen.10 The other report concerns an engineering technician in an analytical chemistry laboratory who had direct skin exposure to aqueous solutions of ninhydrin with concentrations between 2 and 50%."
Petroleum ether
With respect to this evaluation, petroleum ether is used as the carrier agent for both ninhydrin and DFO. Petroleum ether is a fractional distillate of
Page 9 - Health Hazard Evaluation Report No. 92-147
petroleum and consists of a mixture of alkanes. The typical composition of petroleum ether is 80% pentane and 20% isohexane12. Literature specific to the health effects of isohexane is scant. The health hazards of isohexane, as described in the Department of Transportation Emergency Response Guidebook, are similar to the health hazards of both petroleum ether and pentane. 12,13,14 The health effects of pentane are described below.
Pentane
Exposure to pentane may affect the skin, eyes, and nose, and may also decrease alertness. Pentane's solvent properties give it the ability to defat the skin and cause drying. Short-term exposure can produce mild irritation; longer exposure may lead to the development of an irritant dermatitis. Avoidance of exposure combined with thorough washing in case of accidental exposure should prevent these adverse health effects.
Mucous membrane irritation to the eyes and nose may occur
with pentane contact; this contact may be due to splashes or vapor exposure.
When handling liquid pentane, employees should use protective
eye goggles. Contact lenses should not be worn when working with
pentane. An eye-wash station should be available in case of exposure;
the affected eye(s) should be flushed with large amounts of water, occasionally
lifting both upper and lower lids. 1 5 Nasal irritation is usually relieved
by removal from vapors.
Pentane, like many organic solvents, can cause depressant intoxication after short-term, high airborne exposure. Effects may range from decreased alertness to unconsciousness and, at very high exposures, death. In animal experiments, exposures of 130,000 ppm were fatal." Aspiration of pentane will result in direct lung injury, including chemical pneumonitis, pulmonary edema, and hemorrhage. 14 If accidental high exposure occurs, or if toxic symptoms are noted, the individual should be removed to fresh air and supportive measures (e.g., artificial respiration) begun as necessary. Long term neurological effects have not been seen with pentane, but have been demonstrated for many other organic solvents.
Iodine
Iodine is a rare element in nature, found mostly in seawater and geological deposits derived from ancient ocean beds. It is essential for the human thyroid to function properly; deficiency leads to goiter. Common table salt is often fortified with 0.023% potassium iodide as a public health measure to prevent goiter. 1 7 In industry, iodine is used for its oxidizing properties.
Page 10 - Health Hazard Evaluation Report No. 92-147
Medicinal applications include use as an antiseptic, and treatment of thyroid goiter. 18
Iodine is a severe irritant; adverse health effects depend on the route of exposure. Workers may be exposed through inhalation or ingestion; skin or eye exposure can lead to localized irritation.
Inhalation of iodine vapor causes irritation of the respiratory tract; breathing difficulties may have a delayed onset." in severe exposure, pulmonary edema can occur. Iodine is absorbed by the lungs, converted to iodide in the body, and excreted in the urine. 1 7 If high exposure occurs, the individual should be moved to fresh air, and medical attention obtained at once." Artificial respiration may be necessary.
Ingestion of 2-3 grams of iodine may be fatal. In smaller quantities, ingestion can cause a burning sensation in the mouth, vomiting, abdominal pain, and diarrhea. Chronic absorption may cause insomnia, eye and nose inflammation, bronchitis, tremor, rapid heart beat, diarrhea, and weight loss. Employees should not eat or smoke in areas where solid iodine is handled, processed, or stored. Employees handling solid iodine, or liquids containing iodine, should wash their hands thoroughly with soap, or mild detergent, and water prior to eating, smoking, or using toilet facilities.19
Eye exposure to iodine vapor can cause irritation and can stain the cornea brown. A vapor concentration of 0.57 ppm was tolerated by four subjects for 5 minutes without eye irritation; all experienced eye irritation at a concentration of 1.63 ppm after 2 minutes.19 Exposure to vapor saturated with iodine is used in the treatment of dendritic keratitis; the corneal epithelium stains brown and sloughs off. There is complete recovery in 2-3 days."
Workers should not wear contact lenses when using iodine. Eye protection should be used, and an eye-wash station should be available if there is any possibility of exposure to either solid iodine or liquids containing more than 7% iodine by weight. If eye exposure occurs, wash the eyes immediately with large amounts of water, lifting the lower and upper lids occasionally. Medical attention should be obtained immediately."
Crystalline iodine or strong solutions of iodine may cause severe irritation of the skin. It is not easily removed from the skin and may cause burns. An allergic skin rash may occur. Protective clothing and gloves should be used to prevent skin contact with solid iodine or liquid solutions of iodine. Any possible contact with solutions greater than 7% iodine by weight should
Page 11 - Health Hazard Evaluation Report No. 92-147
be avoided; repeated or prolonged contact with solutions less than 7% iodine by weight should be avoided. Clothing which becomes contaminated with iodine should be removed immediately, placed in closed containers for storage, and not worn again until the iodine is removed. If there is any possibility of exposure of an employee's body to solid iodine, or to solutions containing 7% iodine by weight, emergency facilities for quick drenching of the body should be provided within the immediate work area. 1 9
Ethyl-2-cyanoacrylate
Very little toxicity information exists for ethyl-2-cyanoacrylate. Alkyl-2-cyanoacrylate adhesives, as a group, are described as strong irritants,
20
affecting the eyes, nose, and throat. Of these substances, most of the reports concern methyl-2-cyanoacrylate. Studies have indicated an odor threshold for methyl-2-cyanoacrylate ranging from 0.8 to 3.9 ppm. The threshold for throat and nose irritation is reported to be 1.6 to 2.3 ppm, and the threshold for eye irritation and burning about 3.1 ppm. There is an assumption that the other cyanoacrylates (ethyl and butyl) are equally 20
irritating, but they are somewhat less volatile.
Acetone
Acetone is an irritant of the eyes and mucous membranes;
at very high concentrations it is a central nervous system depressant.
21 Acetone is considered to be of low risk to health because few adverse
effects have 22
been reported despite widespread use for many years.
Carbon black
The recommended occupational exposure limits for carbon
black are based on data indicating that it may cause both temporary and
permanent lung damage. NIOSH considers carbon black a potential human
carcinogen.' Particulate polycyclic organic material (PPOM), polynuclear
aromatic hydrocarbons (PNAs), and polycyclic aromatic hydrocarbons (PAHS)
are terms frequently encountered in the literature and often used interchangeably.
Some of these PAHS, such as 3,4-benzpyrene, pyrene, and 1,2-benzpyrene
are formed during carbon black manufacture.
Their adsorption on the carbon black could pose a risk
of cancer after
23
exposure to the carbon black.
Page 12 - Health Hazard Evaluation Report No. 92-147
Polynuclear Aromatic Hydrocarbons (PNAs
PNAs are chemical species that consist of two or more fused aromatic rings. They are often associated with the combustion or pyrolysis of organic matter, especially coal, wood, and petroleum products. Materials associated with these processes have been demonstrated to contain compounds shown to cause cancer in laboratory animals and, in some cases, humans. There are few dose-related relationships for the PNA mixtures that may be found in these processes, and no "safe" exposures to PNA aerosols. Although the application of laboratory animal data for PNA compounds to estimate human risk is very difficult, any occupational exposure to potentially carcinogenic matter is a cause for concern and exposures should be kept to an absolute minimum."
25-28
The potential adverse health effects of PNAs are well
recognized.
Several PNAS, such as benzol(a)anthracene and pyrone,
have been shown to be carcinogenic in animals. A TWA exposure of
0.2 /ug/m' was recommended by the coke oven advisory committee for benzol(a)pyrone
under the OSHA 29 CFR 1910.1029 coke oven emissions standard, but was not
adopted. A special NIOSH hazard review of christen recommended that
it be controlled as an occupational carcinogen. Also, ACGIH includes
christen, benzol(b)fluoranthene, and benzol(a)pyrone
in its list of industrial
29
substances suspected of having carcinogenic potential
in man.
The carcinogenic potential of other PNAs [benzol(a)anthracene,
anthracene,
211
pyrone, and fluoranthenel has also been documented.
Titanium dioxide
Titanium dioxide (Tio2) is a mild pulmonary irritant and is generally regarded as a nuisance dust. 21 Of 15 workers who had been exposed to TiO2 dust, three showed radiographic signs in the lungs resembling "slight fibrosis," but disabling injury did not occur. The magnitude and duration of exposure were not specified.
In an inhalation study on the effects of TiO2, rats were exposed to TiO2 at concentrations of 10, 50, and 250 mg/m' for 6 hours per day, 5 days a week, for 2 years. Rats of both sexes exposed to all concentrations had significant increases in broncho/bronchiolar pneumonia and alveolar cell hyperplasia. Rats of both sexes exposed at the highest dose had a statistically significant increase in bronchioloalveolar adenomas; female rats (1 3 of 74 rats) exposed at the highest dose also had a statistically significant increase in squamous cell carcinomas when compared to non-exposed
1 2
controls (O of 77 rats). These findings meet the OSHA criteria for a
Page 13 - Health Hazard Evaluation Report No. 92-147
potential occupational carcinogen. NIOSH considers titanium dioxide to be a potential human occupational carcinogen and therefore recommends that potential exposure to titanium dioxide be reduced to the lowest feasible level.'
Argon Laser
Lasers produce a special type of light in which the energy is monochromatic and synchronized and the path is highly focused. Risk of acute injury is directly proportional to the power of the laser and the probability of intercepting the laser beam. Although the eye is the most vulnerable site of injury, high-power laser beams can also burn tissue. ACGIH and the American National Standards Institute (ANSI) have developed standards for protecting workers from lasers, but government regulatory agencies have not. 7,33,34
VI. RESULTS
Air Sampling
PBZ and area sample results for ethyl-2-cyanoacrylate, petroleum ether, iodine, acetone, carbon black, and titanium dioxide are presented in Tables 2-9. The NIOSH RELS, ACGIH TLVS, and OSHA PELs for these compounds are presented in Table 1.
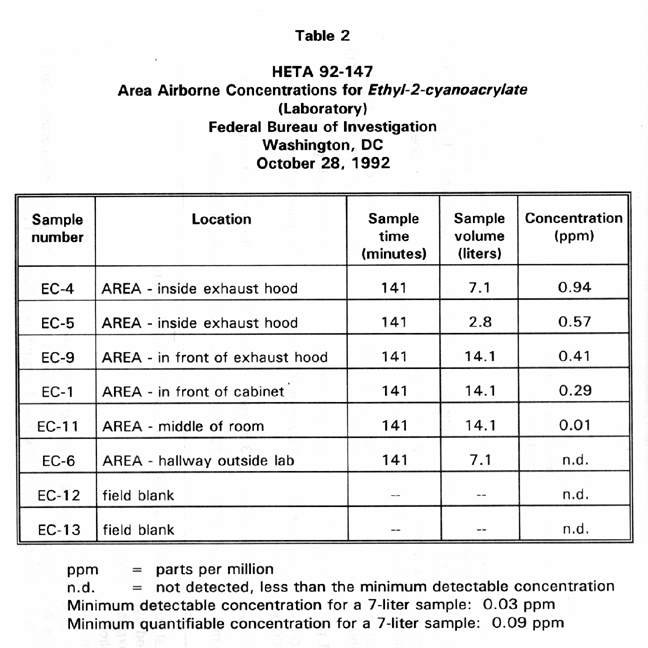
Table 2 presents area sampling results for airborne ethyl-2-cyanoacrylate concentrations in the laboratory while fingerprint specialists used the superglue process to process prints. These concentrations ranged from not detected to 0.94 ppm. The two samples with the highest concentration were collected inside the exhaust hood into which the superglue cabinet was exhausted. Samples E-9 and E-1 were collected in the area in front of the cabinet and exhaust hood. This is the part of the lab where the fingerprint specialists worked. Sample E-1 1 was collected in the middle of the lab. The location of this sample was just beyond the expected cutoff zone of high superglue vapor concentration. This location had been determined using the smoke-generator to reveal air movement throughout the lab.
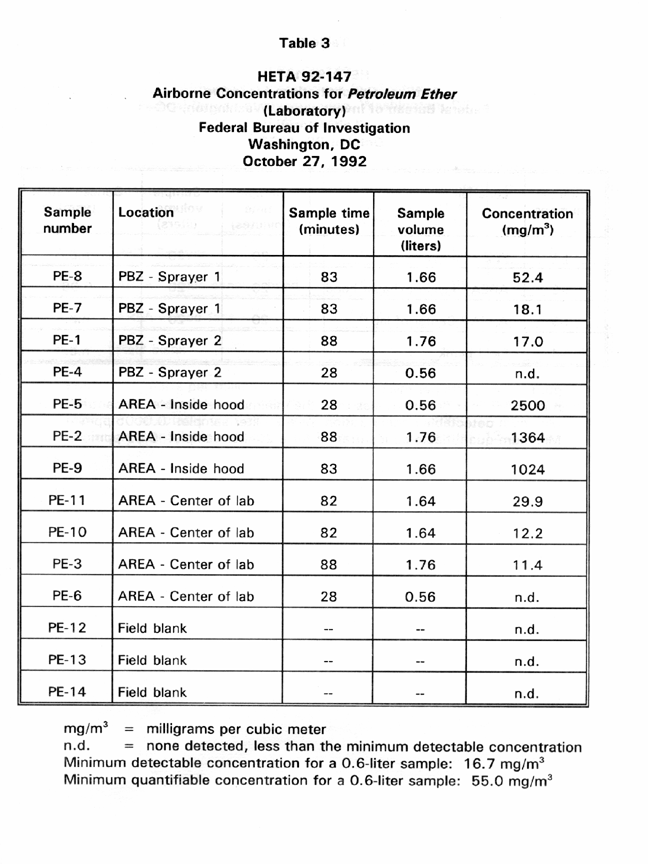
Table 3 presents PBZ and area air sampling results for airborne petroleum ether concentrations in one of the laboratories. PBZ samples ranged in concentration from not detected to 52.4 ppm. Area samples that were collected within the exhaust ranged in concentration from 1024 to 2500 ppm. Area samples collected in the occupied portion of the lab ranged from not detected to 29.9 ppm.
Page 14 - Health Hazard Evaluation Report No. 92-147
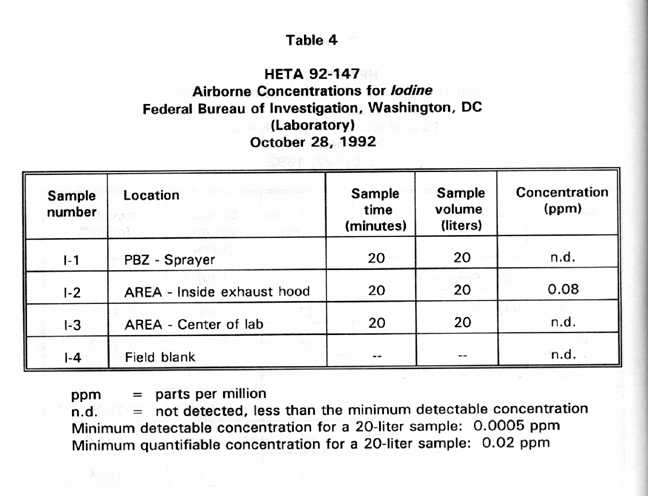
Table 4 presents PBZ and area air sampling results for airborne iodine concentrations for one of the fingerprint specialists using the iodine spray solution in a laboratory exhaust hood. The only sample detecting iodine was the one that was collected inside the exhaust hood. This air sample yielded an airborne iodine concentration of 0.08 ppm.
Table 5 presents PBZ and area air sampling results for airborne acetone concentrations for one of the fingerprint specialists spraying acetone on pieces of evidence in a laboratory exhaust hood. Acetone was detected on the area sample that was collected in the exhaust hood showing an airborne concentration of 111 ppm. Acetone was not detected on the other samples.
Table 6 presents PBZ and area air sampling results for airborne ethyl-2cyanoacrylate concentrations at the simulated crime scene while superglue was used to process latent prints from an automobile. PBZ concentrations for the three fingerprint specialists at the crime scene ranged from 0.24 to 0.54 ppm with a mean of 0.38 ppm. Area samples collected in and around the car ranged in concentration from 0.30 to 0.88 ppm with a mean of 0.61 ppm.
Table 7 presents PBZ and area air sample results for airborne petroleum ether concentrations at the simulated crime scene during use of the ninhydrin petroleum ether solution. PBZ concentrations ranged from 3107 to 5532 Mg/M3 with a mean of 4107 Mg/M3 . Two area airborne concentrations were 723 and 2043 mg/m'.
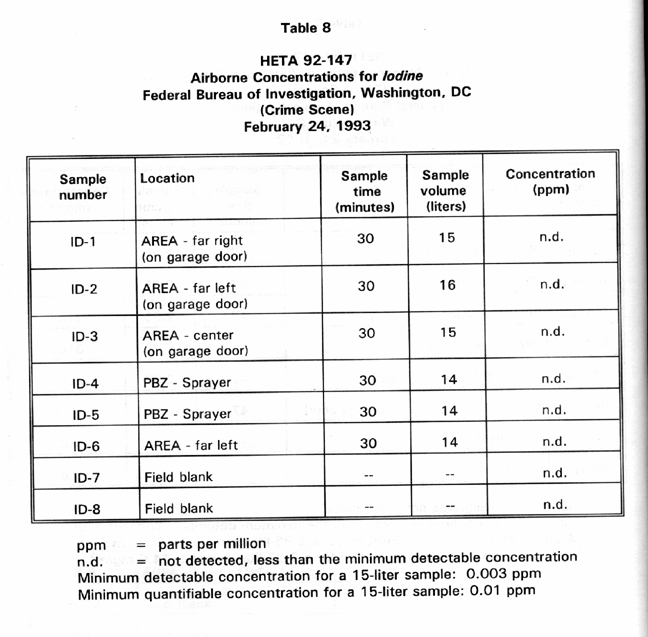
Table 8 presents PBZ and area air sample results for airborne iodine concentrations at the simulated crime scene while a fingerprint specialist used an iodine spray solution to process latent prints from several pieces of wall board. Iodine was not detected on any of the samples.
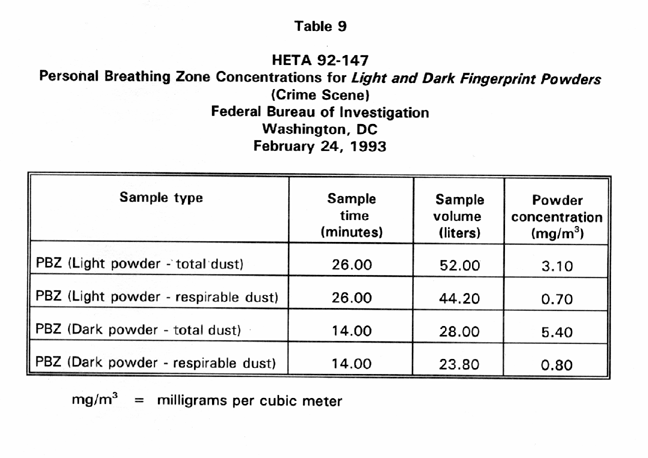
Table 9 presents PBZ air sample results for airborne particulate concentration generated when a fingerprint specialist used fingerprint powders to process latent prints at the simulated crime scene. Respirable dust concentrations for the light and dark powders were low, 0.7 and 0.8 Mg/M3 , respectively. The total particulate concentrations for light and dark powders were higher, 3.1 and 5.4 Mg/M3 , respectively. Two PNAs were detected in trace amounts in a bulk sample of the dark fingerprint powder. Fluoranthene and pyrene were present in the bulk sample in concentrations of 4.2 and 34.0 jig per gram of powder, respectively.
Page 15 - Health Hazard Evaluation Report No. 92-147
Employee Health Interviews
Private interviews were conducted with seven LFPS employees, including one supervisor, in order to identify their concerns about their exposure to potentially hazardous chemicals. In general, persons who experience an unintentional injury at work often become more aware of the hazards associated with work when compared to those people who have not had such an accident. For this reason, employees with a documented history of injury were sought for interview. Three persons were identified through a review of the last six months of the Bureau of Employee Compensation log. This log lists those individuals who suffered a lost-time or compensable injury or illness at work. Two employees were self-identified, conveying to the NIOSH investigators through their union representative that they wished to be interviewed. The two remaining individuals were randomly chosen on the basis of their long tenure in the LFPS. Also, other employees were questioned informally during the investigation regarding current symptoms and future health concerns.
Employees attributed irritant respiratory tract responses, such as coughing, sneezing, and rhinitis (runny nose), to exposures to workplace chemicals, including acetone, and both ninhydrin and DFO in petroleum ether carriers. Symptoms usually occurred in the evidence laboratories, especially when wet evidence was removed from the hoods before being allowed to dry thoroughly. These specialists, intent on doing their job quickly and efficiently, would return evidence to their desks prior to waiting the appropriate amount of time, as protocol demanded, for drying and off-gassing to occur. At these times, symptoms were noted outside of the laboratories. Occasionally, symptoms would occur the day following processing, while the employee was working with evidence at his or her workstation. These symptoms were an occasional and task-specific occurrence.
VII. DISCUSSION
Visual observation of air movement in the laboratories using a smoke generator revealed that all but one of the laboratories were under negative pressure with respect to the rest of the office area. Air was seen to move along the ceiling and out into the hallway from the lab that contained the large superglue cabinet. This was a very slight amount of air movement but it indicated that it is possible for small amounts of airborne contaminants to leave the laboratories and enter the rest of the office space. The exhaust hoods in all of the laboratories were seen to effectively move air from the workers breathing zone into the hoods. Because of this, it is unlikely that
Page 16 - Health Hazard Evaluation Report No. 92-147
airborne chemicals used in the exhaust hoods could move out of the labs and into the office space.
In the laboratory that contained the large superglue cabinet, the smoke generator was used to show how air movement was affected by the cabinet itself. After filling the cabinet with smoke and then allowing about 1 0 minutes for the cabinet to be exhausted, the cabinet doors were opened. Smoke billowed out of the cabinet directly into the worker's breathing zone. The smoke showed that air movement from the cabinet was limited by the adjacent exhaust hood. The smoke moved to about six feet in front of the cabinet, but was then stopped by a combination of the overhead supply air current and the negative pressure from the exhaust hood. Air sampling results for ethyl-2-cyanoacrylate, presented below, supported these visual observations.
There are no environmental evaluation criteria for ethyl-2-cyanoacrylate, so results of air sampling for this substance can only be compared to a similar compound, methyl-2-cyanoacrylate. The OSHA PEL and ACGIH TLV for methyl-2-cyanoacrylate of 2 ppm for an 8-hour day were not surpassed during either the laboratory or simulated crime scene portion of this investigation. However, because of the irritating properties of alkyl-2-cyanoacrylate and the unknown risk that potentially high short-term exposures could cause, the data collected in the lab and at the crime scene are still pertinent. Data from the laboratory show that the ventilation cabinet that was used did not adequately remove superglue vapors from the laboratory. Sample E-1, which was collected in front of the cabinet where the specialists stood, showed an airborne concentration of 0.29 ppm, similar to the PBZ concentrations measured for Specialist C at the crime scene.
The airborne concentration of ethyl-2-cyanoacrylate from
sample E-1 1
(0.01 ppm) confirmed visual observations of air movement
in the laboratory. This sample was collected in the middle of the
laboratory just beyond the reach of air movement from the superglue cabinet.
Sample E-1, which was collected just a couple feet from E-1 1, revealed
a much lower airborne concentration. Although the superglue cabinet
was not effective at removing airborne contaminants from the work area
in the lab, the nearby exhaust hood was powerful enough to contain and
exhaust most of the vapors that were released from the superglue cabinet.
Results of petroleum ether air sampling in the laboratory indicate that
the exhaust hoods effectively removed airborne contaminants from the lab
and from the breathing zone of the fingerprint specialist. Concentrations
inside the hood were approximately the same as area and PBZ concentrations
measured at the crime scene. However, in the laboratory PBZ and area
concentrations
Page 17 - Health Hazard Evaluation Report No. 92-147
from the center of the lab were much less than the concentrations generated in the hood. Although laboratory PBZ concentrations were very low due to the effectiveness of the laboratory exhaust hoods, the crime scene PBZ concentrations were very high because of a lack of ventilation. Air sampling results at the crime scene show that fingerprint specialists can be exposed to potentially hazardous concentrations of petroleum ether at crime scenes.
As with the petroleum ether results, the highest airborne concentration of iodine was measured in the laboratory exhaust hood. Iodine was not detected on any of the other laboratory or crime scene samples, although there is clearly potential for exposure to airborne iodine at the crime scene. Without local exhaust ventilation to remove potentially harmful airborne contaminants from the breathing zones of the specialists, these workers could be potentially exposed to airborne concentrations similar to those measured in the laboratory exhaust hoods.
Results of acetone sampling in the laboratory are similar to the other laboratory air samples. The only sample that had a detectable amount of acetone was the one collected in the exhaust hood. Once again, this indicates that the exhaust hoods effectively removed airborne contaminants from the laboratory and from the breathing zone of the fingerprint specialist.
Some problems with the operation of two lasers were observed. Evidence that arrived at the LFPS was opened in the laser room and examined using the lasers to detect fingerprints. Many safety precautions are necessary when using such a potentially hazardous light source. The protective goggles that were used by the specialists were not labeled properly according to the ANTI standard. Some of the lenses were cracked, and in some cases there were different types of filters in the same pair of goggles. The door to the laser room had a small window in it, but there was no filter for the window. Appropriate laser warning signs were not on any doors or walls. The warning light in the hallway outside of the laser room was on throughout the day because the laser was always turned on, even though the laser was not always being used. The workers using the lasers were unaware of many of the hazards associated with using an argon laser.
Although air samples were not collected in the LFPS offices, it is likely that very low levels of the chemicals used in the laboratory are present in the office area. Because the labs are under negative pressure, contaminants would not likely enter the offices by air movement from the labs. However, after spraying the evidence the fingerprint specialists sometimes carry it to their desks for examination before it is allowed to dry sufficiently. Also,
Page 18 - Health Hazard Evaluation Report No. 92-147
chemicals from processing could off-gas while the evidence is stored in the many cabinets throughout the LFPS. Laboratory exhaust hoods are ducted directly out of the building, but any contaminants that are introduced to the office air can be recirculated throughout the LFPS.
The total airborne particulate concentration of the dark fingerprint powder (essentially carbon black) at the crime scene was higher than the NIOSH REL of 3.5 Mg/M3 during the 14-minute sample period. The 8-hour or 10-hour TWA would therefore be considerably less than the REL. Specialists using dark fingerprint powders should be aware of the possible presence of PNAs which are present in trace amounts from the production of carbon black. The MSDS or the manufacturer should be able to provide information concerning the presence of PNAS.
Besides developing latent fingerprints, the fingerprint specialists also mix the chemicals and reagents that are used for their work. This includes mixing many potentially hazardous liquid and powdered chemicals. In many cases, the exhaust hoods are not used during mixing of chemicals and powders. Many of the chemicals were stored in cabinets in the exhaust hoods. Such an arrangement impedes the effectiveness of the hoods and is improper for chemical storage. Personal protective equipment used by the specialist included various types of gloves to avoid dermal contact with the many chemicals they handled, and dust masks for when they used fingerprint powders. Some specialists did not always wear gloves because it impeded their ability to work effectively.
VIII. RECOMMENDATIONS
The following recommendations are presented to help reduce the potential for exposure to chemicals used by FBI fingerprint specialists in the laboratory and at crime scenes. These recommendations are based on the environmental sampling results and observations made during the sampling surveys.
1. The large metal cabinet that was used for the superglue process should be replaced with a properly designed and ventilated chamber. This would reduce the potential for exposure to potentially high concentrations of ethyl-2-cyanoacrylate when the cabinet is opened.
2. Because local exhaust ventilation is not available at crime scenes, fingerprint specialists are often exposed to high short-term airborne concentrations of ethyl-2-cyanoacrylate, iodine, and other hazardous chemicals. While using the superglue process or iodine spray at crime
Page 19 - Health Hazard Evaluation Report No. 92-147
scenes, fingerprint specialists should use respirators that provide maximum protection. Because of the unknown hazard associated with ninhydrin, maximum respiratory protection should be used when using the ninhydrin solution at crime scenes. The light fingerprint dusting powder contains titanium dioxide and the dark powder contains carbon black, both of which are considered by NIOSH to be potential human carcinogens. NIOSH recommends that worker exposure to potential human carcinogens should be kept to the lowest feasible limit. Respirator options when using any of these chemicals at the crime scene or any other unventilated area include: (1) a self-contained breathing apparatus (SCBA) that has a full facepiece and is operated in a positive-pressure mode, or (2) a supplied-air respirator that has a full facepiece and is operated in a pressure demand or other positive-pressure mode in combination with an auxiliary SCBA operated in a pressure-demand or other positive-pressure mode.
3. Because of the possibility of generating airborne concentrations of carbon black which are in excess of the established occupational exposure criteria, fingerprint specialists should use respirators with high efficiency particulate air filters (HEPA) when using the dark fingerprint powder at a crime scene.
4. Several improvements could be made to reduce the risk of hazard or injury due to improper use of the argon laser.
+ All users of the laser should be provided with a training
course in
laser safety, including the use of protective equipment.
+ Proper eye protection should be provided and maintained for laser operators.
+ The window in the door to the laser room should be provided with a .protective filter or a new door without a window should be installed.
+ The laser should be turned off when it is not being
used.
This would reduce the possibility of accidental exposure
to anyone entering the laser room. This would also allow the warning
light to warn people not to enter the laser room.
5. To avoid low level contamination of the office air, fingerprint specialists should allow all pieces of evidence to dry thoroughly before removing them from the exhaust hoods.
Page 20 - Health Hazard Evaluation Report No. 92-147
6. To further reduce office air contamination, evidence cabinets should be located in a room that is under negative pressure and that has a dedicated exhaust system.
7. Fingerprint specialists should not use their desks to examine evidence that has been chemically processed. Because they often eat and drink at their desks, they increase the risk of chemical exposure due to ingestion. Evidence examination rooms that are under negative pressure and which have dedicated exhaust systems should be provided.
8. Chemicals should not be stored in the laboratory exhaust hoods. They should be stored in cabinets designed for the storage of flammable and combustible liquids.
9. All mixing of chemical solutions, powders, and other reagents should take place in the exhaust hoods. Because some spray solutions are very difficult to prepare in the existing hoods, exhaust hoods with lower counter tops should be provided for mixing these solutions.
10. In indoor workplaces where smoking is permitted, environmental tobacco smoke (ETS) can spread throughout the airspace of all workers. The most direct and effective method of eliminating ETS from the workplace is to prohibit smoking in the workplace. Until that is achieved, employers can designate separate, enclosed areas for smoking, with separate ventilation systems. Air from this area should be exhausted directly outside the building and not recirculated within the building or mixed with the general dilution ventilation for the
15
building.
11. If manpower resources are available, it would benefit the FBI to have an industrial hygienist on their staff to more thoroughly evaluate, assess, and ultimately reduce or eliminate the potential hazards to which fingerprint specialists are exposed.
IX. REFERENCES
1. OSHA [19851. Methyl-2-cyanoacrylate, ethyl-2-cyanoacrylate: method no. 55 (issued October, 1985). OSHA manual of analytical methods. Salt Lake City, UT: U.S. Department of Labor, Occupational Safety and Health Administration.
Page 21 - Health Hazard Evaluation Report No. 92-147
2. NIOSH [19841. Naphthas: method no. 1550.
In: Eller PM, ed.
NIOSH manual of analytical methods. 3rd rev. ed.
Cincinnati, OH: U.S. Department of Health and Human Services, Public Health
Service, Centers for Disease Control, National Institute for Occupational
Safety and Health, DHHS (NIOSH) Publication No. 84-100.
3. NIOSH [19871. Iodine: method no. 6005 (supplement
issued 8/15/87). In: Eller PM, ed. NIOSH manual of analytical
methods.
3rd rev. ed. Cincinnati, OH: U.S. Department of
Health and Human Services, Public Health Service, Centers for Disease Control,
National Institute for Occupational Safety and Health, DHHS (NIOSH) Publication
No. 84-1 00.
4. NIOSH [19891. Ketones 1: method no. 1300 (supplement
issued 5/15/89). In: Eller PM, ed. NIOSH manual of analytical
methods.
3rd rev. ed. Cincinnati, OH: U.S. Department of
Health and Human Services, Public Health Service, Centers for Disease Control,
National Institute for Occupational Safety and Health, DHHS (NIOSH) Publication
No. 84-100.
5. NIOSH [19841. Nuisance dust, total: method no. 0500. In: Eller PM, ed. NIOSH manual of analytical methods. 3rd rev. ed. Cincinnati, OH: U.S. Department of Health and Human Services, Public Health Service, Centers for Disease Control, National Institute for Occupational Safety and Health, DHHS (NIOSH) Publication No. 84-100.
6. NIOSH [ 1 9921. Recommendations for occupational
safety and health. Cincinnati, OH: U.S. Department of Health and
Human Services, Public Health Service, Centers for Disease Control, National
Institute for Occupational Safety and Health, DHHS (NIOSH) Publication
No. 92-1 00.
7. ACGIH [19931. Threshold limit values for chemical substances and physical agents and biological exposure indices 1993-1994. Cincinnati, OH: American Conference of Governmental Industrial Hygienists.
8. Code of Federal Regulations [ 1 9921. OSHA: Table
Z- 1. 29 CFR
1910.1000. Washington, DC: U.S. Government Printing Office,
Federal Register.
9. Nemeroff NH [ 1 991 1. Ninhydrin and ninhydrin analogs: syntheses and applications. Tetrahedron 47(42):8791-8830.
Page 22 - Health Hazard Evaluation Report No. 92-147
10. Schlacke KH, Fuchs T [ 1 9891. Allergic contact eczema to ninhydrin [German]. Dermatosen In Beruf Und Umwelt - Occupational & Environmental Dermatoses 37(5):179-80.
11. KVstaubaeva KS, Shaparenko MV [ 1 974]. A case of occupational eczema caused bV ninhydrin [Russian]. Gigiena Truda i Professioanl'nVe Zabolevaniia 18(6):46-47.
12. Hazardous Substances DataBank [ 1 9921. Number 2892 petroleum ether. Revision date 4/16/92.
13. Hazardous Substances DataBank [ 1 9901. Number 1125 2-methylpentane. Revision date 10/10/90.
14. Hazardous Substances DataBank [ 1 9921. Number 109 pentane. Revision date 4/27/92.
15. NIOSH/OSHA [1 9781. Occupational health guidelines
for pentane.
In: Mackison FW, Stricoff RS, Partridge LJ, Jr., eds.
NIOSH/OSHA Occupational health guidelines for chemical hazards. U.S.
Department of Health and Human Services, Public Health Service, Centers
for Disease Control, National Institute for Occupational Safety and Health.
U.S. Department of Labor, Occupational Safety and Health Administration,
DHHS (NIOSH) Publication No. 81-123.
16. Gerr F, Letz R [19921. Solvents. In: Rom W, ed. Environmental and occupational medicine, 2nd ed. Boston, MA: Little, Brown.
17. Parmeggiani L, ed. [19831. Iodine. In:
Parmeggiani L, ed.
Encyclopedia of occupational health and safety, 3rd (revised)
ed.,
Geneva: International Labour Office.
18. Hazardous Substances DataBank [ 1 992]. Number 34 iodine. Revision date 4/27/92.
19. NIOSH [1 9781. Occupational health guidelines for iodine. In: Mackison FW, Stricoff RS, Partridge LJ, Jr., eds. NIOSH/OSHA Occupational health guidelines for chemical hazards. U.S. Department of Health and Human Services, Public Health Service, Centers for Disease Control, National Institute for Occupational Safety and Health. U.S. Department of Labor, Occupational Safety and Health Administration, DHHS (NIOSH) Publication No. 81-123.
Page 23 - Health Hazard Evaluation Report No. 92-147
20. United Rubber Workers Journal [ 1 9831. April, 1 983.
21. Hathaway GJ, Proctor NH, Hughes JP, Fischman ML [19911. Chemical hazards of the workplace. 3rd ed. New York, NY: Van Nostrand Reinhold Company.
22. NIOSH [ 1 9781. Criteria for a recommended standard: occupational exposure to ketones. Cincinnati, OH: National Institute for Occupational Safety and Health, DHEW (NIOSH) Publication No. 78-173.
23. NIOSH [19781. Criteria for a recommended standard: occupational exposure to carbon black. Cincinnati, OH: National Institute for Occupational Safety and Health, DHEW (NIOSH) Publication No. 78-204.
24. NIOSH [19911. Hazard evaluation and technical assistance report: Armco Coke Oven, Ashland, KY. Cincinnati, OH: U.S. Department of Health and Human Services, Public Health Service, Centers for Disease Control, National Institute for Occupational Safety and Health, NIOSH Report No. HETA 88-377-2120.
25. Harrison's Principles of Internal Medicine [19801. Ninth Edition. New York, NY: McGraw-Hill Book Company, pp 255-262.
26. NIOSH [ 1 978). Criteria for a recommended standard: occupational exposure to coal-tar products. Cincinnati, OH: U.S. Department of Health, Education, and Welfare, Public Health Service, Center for Disease Control, National Institute for Occupational Safety and Health, DHEW (NIOSH) Publication No. 78-107.
27. Herwin RL, Effect EA [19761. Health Hazard Evaluation Report No. 75194-324. Cincinnati, OH: U.S. Department of Health, Education, and Welfare, Public Health Service, Center for Disease Control, National Institute for Occupational Safety and Health.
28. Gorman RW, Liss GM [ 1 981 1. Health Hazard Evaluation Report No. 80206-1164. Cincinnati, OH: U.S. Department of Health and Human Services, Public Health Service, Centers for Disease Control, National Institute for Occupational Safety and Health.
29. ACGIH [19911. Notice of intended changes - benzo(b)fluoranthene. Appl Occup and Env Hyg 6:805-807.
Page 24 - Health Hazard Evaluation Report No. 92-147
30. Browning E [ 1 9691. Toxicity of industrial
metals. 2nd ed.
London: Butterworths.
31. AIHA [1 9781. AIHA hygienic guide series: titanium dioxide. Akron, OH: American Industrial Hygiene Association.
32. Lee KP, Trochimowicz HF, Reinhardt CF [19851. Pulmonary response of rats exposed to titanium dioxide (Tio2) by inhalation for two years. Toxicol Appi Pharmacol 79:179-192.
33. Weeks JL, Levy BS, Wagner GR, eds. [ 1 991 1. Preventing
occupational disease and injury. American Public Health Association,
Washington DC: Reese Press, Inc.
34. ANSI [ 1 9861. ANSI Standard for the safe use of lasers, Z 1 36. 1, New York. NY: American National Standards Institute.
35. NIOSH [ 1 991 1. Current intelligence bulletin 54:
Environmental tobacco smoke in the workplace - lung cancer and other health
effects. Cincinnati, OH: U.S. Department of Health and Human Services,
Public Health Service, Centers for Disease Control, National Institute
for Occupational Safety and Health, DHHS (NIOSH) Publication No. 91-
108.
X. AUTHORSHIP AND ACKNOWLEDGEMENTS
Report Prepared by: Michael E. Barsan
Industrial Hygienist
Industrial Hygiene Section
Faye T. Bresler, M.D.
Medical Officer
Medical Section
Report Formatted by: Ellen Blythe
Office Automation Assistant
Field Assistance: Eric Esswein
Industrial Hygienist
Dino Mattorano
Industrial Hygienist
Page 25 - Health Hazard Evaluation Report No. 92-147
Angela Weber
Industrial Hygienist
Eugene Moss
Health Physicist
Originating Office:
Hazard Evaluations and Technical
Assistance Branch
Division of Surveillance, Hazard
Evaluations and Field Studies
XI. DISTRIBUTION AND AVAILABILITY
Copies of this report may be freely reproduced and are not copyrighted. Single copies of this report will be available for a period of 90 days from the date of this report from the NIOSH Publications Office, 4676 Columbia Parkway, Cincinnati, Ohio 45226. To expedite your request, include a self-addressed mailing label along with your written request. After this time, copies may be purchased from the National Technical Information Service (NTIS), 5285 Port Royal Road, Springfield, Virginia 22161. Information regarding the NTIS stock number may be obtained from the NIOSH Publications Office at the Cincinnati address.
Copies of this report have been sent to:
1 - Section Chief, FBI Latent Fingerprint Section
2. Assistant Section Chief, FBI Latent Fingerprint Section
3. Occupational Health Nurse, FBI Latent Fingerprint
Section
4. OSHA (Region 111)
For the purpose of informing affected employees, copies of this report shall be posted by the employer in a prominent place accessible to the employees for a period of 30 calendar days.